Chlorate ion bonding6/19/2023 
This indicates that chlorine has undergone both oxidation and reduction in this reaction, making it an example of a disproportionation reaction. In HCl, chlorine has an oxidation state of -1, and in HClO, chlorine has an oxidation state of +1. In Cl2, chlorine has an oxidation state of +0. Let's examine the oxidation states of chlorine in the three different species. The equation for the reaction is as follows: This technique is used to disinfect water for swimming pools and drinking purposes. It is a potent oxidizing agent that can kill various bacteria and viruses, ranging from the common cold to cholera. Chloric acid is also known as hypochlorous acid and is based on the chlorate ion. When chlorine reacts with water, it produces a mixture of hydrochloric acid (HCl) and chloric acid (HClO). How does chlorine react with water?Ĭhlorine plays many roles in everyday life, but we’re going to focus specifically on one of its main uses: treating wastewater. 
If you're having trouble understanding oxidation states, head over to Redox for more information. In chlorate(V), chlorine has an oxidation state of +5. The Roman numeral indicates the oxidation state of one of the elements in a compound. When people refer to chlorate, they usually mean chlorate(V), which is a different ion with the formula. 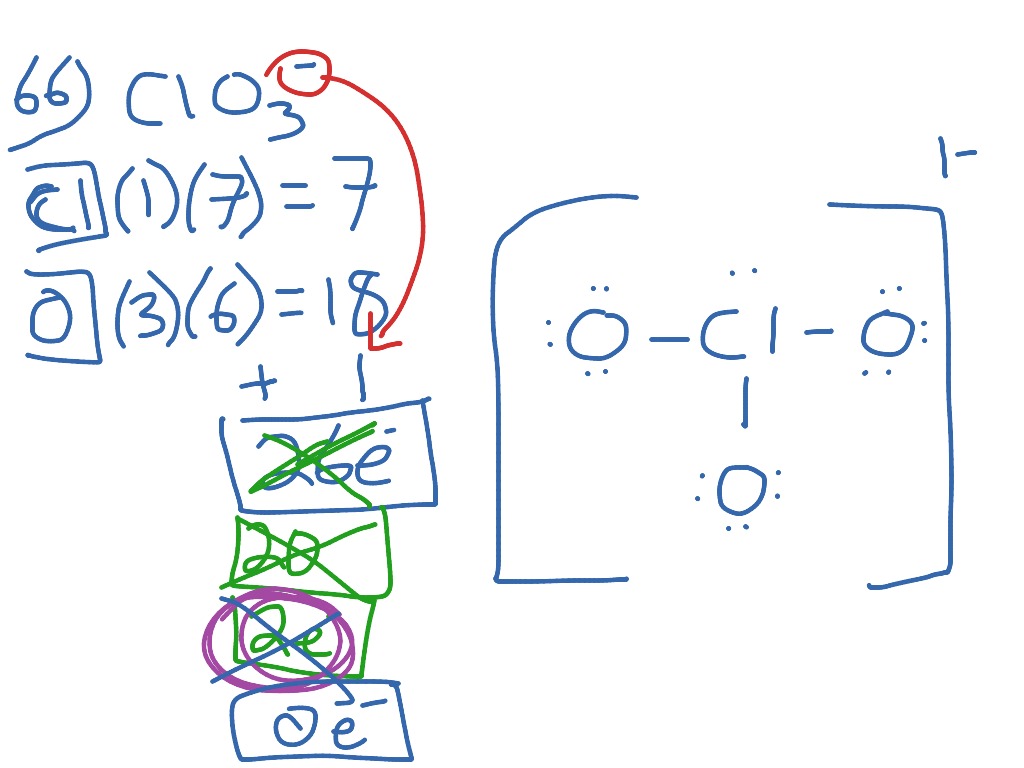
This may seem unusual because chlorine typically has an oxidation state of -1, but in this compound, it takes a higher oxidation state because it is less electronegative than oxygen. In order for all the oxidation states in the compound to equal -1, chlorine has an oxidation state of +1. Oxygen is the most electronegative element and therefore has an oxidation state of -2 (except in some cases). Within the compound, the element with the highest electronegativity takes the lowest oxidation state. In this case, all the oxidation states should add up to -1. In an ion, the sum of all the oxidation states equals the charge on the ion. Let's examine the oxidation states within the compound. Each atom within the molecule has an oxidation state of +0, and the molecule itself is neutral. At room temperature, it appears as a yellow gas made up of two chlorine atoms that are covalently bonded together to form a single molecule. ChlorineĬhlorine is a type of halogen that can be found in group 17 of the periodic table. Despite their similar names, they do have their differences and you shouldn’t get them mixed up. What are chlorine, chloride, and chlorate(I)?īefore we go any further, we need to look at some of the species related to chlorine: chloride and chlorate(I). Learn about the physical chemistry of chlorine reactions in this article. Lastly, we will discuss the advantages and disadvantages of using chlorine in treating wastewater. We will also look at how chlorine reacts with oxygen, sodium, and other halide solutions. Then, we will talk about how chlorine and chlorate(I) are used to clean wastewater and the reaction between chlorine and sodium hydroxide. First, we will explain the differences between chlorine, chloride, and chlorate(I). In this article, we will be discussing the different reactions of chlorine and its compounds. Chlorine is a type of element that belongs to group 17 on the periodic table.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
